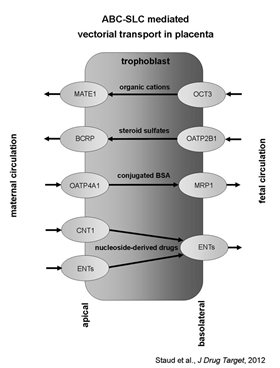
 (excerpted from Staud et al., 2012)
(excerpted from Staud et al., 2012)
In the transfer of drugs across the placenta, both passive diffusion and transporter-facilitated forms of transports are involved. The majority of clinically used drugs are lipid-soluble molecules, thus passive diffusion is the most common way of their transplacental passage. However, with the accumulating evidence on drug transporters in the placenta and the number of their recognized substrates and inhibitors, it is evident that placental transport proteins play a critical role in materno-fetal disposition of drugs. The net placental transport is then given by combination of passive diffusion and transporter-mediated transfer.
To quantify transplacental passage of drugs and to evaluate the role of drug transporters in this event, we have recently proposed a pharmacokinetic model that discriminates between passive diffusion and transporter-mediated transport across the placenta (Staud et al., 2006; Cygalova et al., 2009).
Passive diffusion of a drug across the placenta is typically governed by Fick´s law and, therefore, depends on physical-chemical properties of the molecule, surface area and thickness of the placenta, and transplacental concentration gradient. On the other hand, transporter-mediated transfer is a capacity-limited event that can be described by Michaelis-Menten kinetics. Both types of transport can be expressed by means of drug clearance, that is, clearance of passive diffusion (Clpd) and clearance of transporter-mediated process (Cltm). As clearance is an additive parameter, the total transplacental clearance is a combination of Clpd and Cltm. For example, P-glycoprotein-mediated efflux in the placenta runs exclusively from fetus to mother. Therefore, total transplacental clearance of a lipid soluble substrate of P-glycoprotein in the fetal-to-maternal direction (Clfm) will be calculated by adding the two particular clearances as follows:
Clfm = Clpd + Cltm eq. 1
On the other hand, in the maternal-to-fetal direction (Clmf), P-glycoprotein-mediated transport is subtracted from passive diffusion:
Clmf = Clpd - Cltm eq. 2
In the clearance concept, transplacental passage mediated by a transport protein can be expressed as follows:
Cltm = Vmax/(Km + Cma(fa)) eq. 3
where Vmax is the maximal velocity of the transport, Km is the concentration at which half the maximal velocity is reached, and Cma(fa) is substrate concentration in the maternal (Cma) or fetal (Cfa) circulation. Adding equation 3 to 1 and 2 yields the final formulas describing total transplacental clearance in fetal to maternal (Clfm) and maternal-to-fetal (Clmf) directions:
Clfm = Clpd + Vmax/(Km + Cma(fa)) eq. 4
and
Clmf = Clpd - Vmax/(Km + Cma) eq. 5
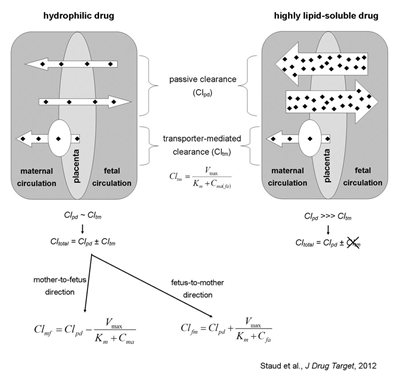 Role of efflux transporters in drug transport across the placenta; effect of lipid-solubility. Transport of drugs across the placenta is a function of passive and transporter-mediated processes. ABC efflux transporters are mainly localized to the apical, mother-facing membrane and pump their substrates from placenta to maternal circulation. These transport proteins, therefore, limit mother-to-fetus and accelerate fetus-to-mother transport of xenobiotics, thus, forming functional part of the placental barrier. In the case of hydrophilic/charged compounds, the role of passive diffusion is rather limited and the transporter-mediated clearance plays a great role in the transplacental pharmacokinetics. On the other hand, lipid solubility may overshadow the role of active transport; consequently, highly lipophilic compounds are transported mainly by passive diffusion and their transplacental pharmacokinetics is not significantly affected by placental efflux transporters. See text for explanation.
Role of efflux transporters in drug transport across the placenta; effect of lipid-solubility. Transport of drugs across the placenta is a function of passive and transporter-mediated processes. ABC efflux transporters are mainly localized to the apical, mother-facing membrane and pump their substrates from placenta to maternal circulation. These transport proteins, therefore, limit mother-to-fetus and accelerate fetus-to-mother transport of xenobiotics, thus, forming functional part of the placental barrier. In the case of hydrophilic/charged compounds, the role of passive diffusion is rather limited and the transporter-mediated clearance plays a great role in the transplacental pharmacokinetics. On the other hand, lipid solubility may overshadow the role of active transport; consequently, highly lipophilic compounds are transported mainly by passive diffusion and their transplacental pharmacokinetics is not significantly affected by placental efflux transporters. See text for explanation.
In the case of drug efflux transporters, it can be assumed that these will show noticeable effect in transplacental pharmacokinetics and fetal protection only if the rate of Cltm is considerable in comparison to Clpd. If the drug is highly lipid soluble, Clpd will be substantially larger than Cltm and, subsequently, transporter activity will be overwhelmed by the passive diffusion and will have negligible effect on transplacental pharmacokinetics (see figure). We have recently confirmed this assumption in a study using various P-glycoprotein and BCRP substrates; we observed that a rise in lipid solubility increases the passive diffusion and, at the same time, decreases the efflux transporter effectiveness. Even in the case of BODIPY FL prazosin, a dual substrate of both P- glycoprotein and BCRP, the combined effect of both transporters was suppressed by high lipid solubility of the molecule and, therefore, rapid clearance by passive diffusion (Cygalova et al., 2009). On the other hand, placental transport of drugs that show slow passive diffusion will be greatly affected by transporter activity and, therefore, is prone to transporter malfunctioning, such as inhibition, down-regulations, drug interactions or polymorphisms.
Literature:
Cygalova LH, Hofman J, Ceckova M and Staud F (2009) Transplacental pharmacokinetics of glyburide, rhodamine 123, and BODIPY FL prazosin: effect of drug efflux transporters and lipid solubility. J Pharmacol Exp Ther 331:1118-1125.
Staud F, Vackova Z, Pospechova K, Pavek P, Ceckova M, Libra A, Cygalova L, Nachtigal P and Fendrich Z (2006) Expression and transport activity of breast cancer resistance protein (Bcrp/Abcg2) in dually perfused rat placenta and HRP-1 cell line. J Pharmacol Exp Ther 319:53-62.
Staud F, Cerveny L and Ceckova M (2012) Pharmacotherapy in pregnancy; effect of ABC and SLC transporters on drug transport across the placenta and fetal drug exposure. J Drug Target 20:736-763.